What is Propane?
Propane was first identified by Dr.Walter O Snelling of the United States of America Bureau of Mines in 1910, he discovered a energy-rich gas that is related to petroleum and natural gas. Propane is know as C3H8 and is therefore formed with 3 Carbon atoms & 8 Hydrogen atoms, propane is classified as a hydrocarbon and is often referred to as LP gas. Snelling studied the process of separating "wild gasoline's" into both liquid & gaseous states ( the volatility meaning the ability of a substance to vaporize was high making light hydrocarbons know as wild gasoline's). Through his research he discovered a gas that could be stored in a liquid state & be used to power houses for almost 3 weeks, which was a huge advancement towards what we know as modern day uses for propane and from then on the gas was refined and the creation of American Gasoline Co occurred shortly after which was the first marketer of propane. By 1947, 62% of U.S homes had been equipped with propane for cooking, also know as the modern day propane grill.
Propane was first identified by Dr.Walter O Snelling of the United States of America Bureau of Mines in 1910, he discovered a energy-rich gas that is related to petroleum and natural gas. Propane is know as C3H8 and is therefore formed with 3 Carbon atoms & 8 Hydrogen atoms, propane is classified as a hydrocarbon and is often referred to as LP gas. Snelling studied the process of separating "wild gasoline's" into both liquid & gaseous states ( the volatility meaning the ability of a substance to vaporize was high making light hydrocarbons know as wild gasoline's). Through his research he discovered a gas that could be stored in a liquid state & be used to power houses for almost 3 weeks, which was a huge advancement towards what we know as modern day uses for propane and from then on the gas was refined and the creation of American Gasoline Co occurred shortly after which was the first marketer of propane. By 1947, 62% of U.S homes had been equipped with propane for cooking, also know as the modern day propane grill.
How does Propane work?
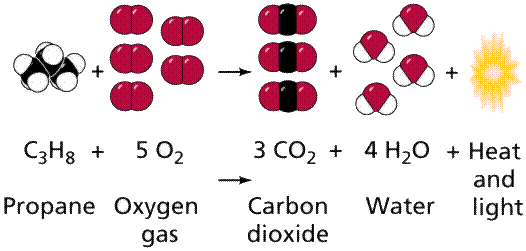
Propane is used in cooking due to the fact that when the gas is exposed to oxygen it is able to produce heat which we use to cook our food, which is therefore the function of a propane grill. This type of reaction is called a complete combustion reaction. The balanced equation for this reaction is as follows C3H8 + 5 O2 ------> 3 CO2 + 4 H2O + heat. This reaction is able to take place due to the fact that there is air (oxygen) surrounding our outdoor grills and a fuel source the propane tank which then combine with the pre-existing heat source of a grill lighter to utilize its thermal energy to created fire at the bottom of the grill which is the heat source which is used for the cooking, because of the heat the water has then changed states to be present in the form of steam and the CO2 gas is released back into the environment.
Propane is used in cooking due to the fact that when the gas is exposed to oxygen it is able to produce heat which we use to cook our food, which is therefore the function of a propane grill. This type of reaction is called a complete combustion reaction. The balanced equation for this reaction is as follows C3H8 + 5 O2 ------> 3 CO2 + 4 H2O + heat. This reaction is able to take place due to the fact that there is air (oxygen) surrounding our outdoor grills and a fuel source the propane tank which then combine with the pre-existing heat source of a grill lighter to utilize its thermal energy to created fire at the bottom of the grill which is the heat source which is used for the cooking, because of the heat the water has then changed states to be present in the form of steam and the CO2 gas is released back into the environment.
Reactants & Products...
The components of any chemical reaction are the reactants the materials you start with in this case the reactants are Propane gas and oxygen, then there are also the products the substances that are created as a result of the reaction which in this case are water, carbon dioxide and heat. Each of theses substances are unique and have there own chemical & physical properties, for example propane gas has a melting point of -187.7 C & a boiling point of -42.1 C & a flash point of -103.4 C, and flammable limits of 2.4%-9.5% & a heat of combustion of -2220.1 KJ/mole . While oxygen has a boiling point of -183 C and a melting point of -219 C & oxygen is highly combustible & oxygen is highly reactive with group one & two metals & oxygen has two allotropes. Whereas water has a boiling point of 100 C & a melting point of 0 C & a pH level of 7, is non-flammable & pure water is toxic. Carbon Dioxide has a boiling point of -78.4 C & a melting point of -56.6 C & an electrical discharge will cause CO2 to decompose into carbon monoxide & oxygen, CO2 will combine with water(vapor or liquid) to from carbonic acid & CO2 reacts with group 2 metals to give carbonates & bicarbonates.
The components of any chemical reaction are the reactants the materials you start with in this case the reactants are Propane gas and oxygen, then there are also the products the substances that are created as a result of the reaction which in this case are water, carbon dioxide and heat. Each of theses substances are unique and have there own chemical & physical properties, for example propane gas has a melting point of -187.7 C & a boiling point of -42.1 C & a flash point of -103.4 C, and flammable limits of 2.4%-9.5% & a heat of combustion of -2220.1 KJ/mole . While oxygen has a boiling point of -183 C and a melting point of -219 C & oxygen is highly combustible & oxygen is highly reactive with group one & two metals & oxygen has two allotropes. Whereas water has a boiling point of 100 C & a melting point of 0 C & a pH level of 7, is non-flammable & pure water is toxic. Carbon Dioxide has a boiling point of -78.4 C & a melting point of -56.6 C & an electrical discharge will cause CO2 to decompose into carbon monoxide & oxygen, CO2 will combine with water(vapor or liquid) to from carbonic acid & CO2 reacts with group 2 metals to give carbonates & bicarbonates.
What are all the uses for Propane?
Propane has several uses one of the most common being used for cooking on propane grills, also pool & hot tub heaters, patio heaters, furnaces (heating homes another huge use of propane), hot air balloon fuel, fireplaces & power generators.
Propane has several uses one of the most common being used for cooking on propane grills, also pool & hot tub heaters, patio heaters, furnaces (heating homes another huge use of propane), hot air balloon fuel, fireplaces & power generators.
Safety when working with Propane...
When handling propane there are a few very important safety procedures that must be followed to ensure you are handling propane safely. Propane is stored in a steel cylinder which is a high pressure system and therefore makes propane highly flammable and explosive. This means that propane must be stored away from any heat source and in the proper orientation to ensure the pressure relieve valve is working in-case of overfill and the exposure to excessive heat. Furthermore propane must be stored outdoors in case of leak and away from any possible heat source or opportunity of possible ignition.
When handling propane there are a few very important safety procedures that must be followed to ensure you are handling propane safely. Propane is stored in a steel cylinder which is a high pressure system and therefore makes propane highly flammable and explosive. This means that propane must be stored away from any heat source and in the proper orientation to ensure the pressure relieve valve is working in-case of overfill and the exposure to excessive heat. Furthermore propane must be stored outdoors in case of leak and away from any possible heat source or opportunity of possible ignition.
The balanced chemical equation of the complete combustion reaction that powers
a propane grill.
a propane grill.
Dr. Walter. O Snelling